Application
These Secondary Standards are qualified as Certified Reference Materials. These are suitable for use in several analytical applications including but not limited to pharma release testing, pharma method development for qualitative and quantitative analyses, food and beverage quality control testing, and other calibration requirements.
General description
Pharmaceutical secondary standards for application in quality control, provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards. Calcium Ascorbate is a non-acidic buffered form of ascorbate, which is found to be safer for mouth and sensitive stomach. It can also be directly applied to gums to heal infections.[1]
Other Notes
To see an example of a Certificate of Analysis for this material enter LRAC3786 in the slot below. This is an example certificate only and may not be the lot that you receive.
Calcium Ascorbate
Pharmaceutical Secondary Standard; Certified Reference Material
Code: PHR1361
- Manufacturer: Merck
- Trademark: Supelco
- Hãng sản xuất: Merck
Kiểm tra giá Calcium Ascorbate
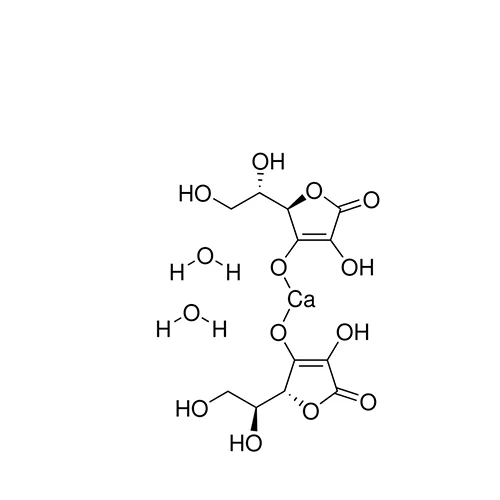
Calcium Ascorbate
Bạn vui lòng nhập đúng số điện thoại để chúng tôi sẽ gọi xác nhận đơn hàng trước khi giao hàng. Xin cảm ơn!


 Tiếng Việt
Tiếng Việt