Analysis Note
These secondary standards offer multi-traceability to the USP and EP primary standards, where they are available.
Application
Rufinamide may be used as a pharmaceutical reference standard for the quantification of the analyte in pharmaceutical formulations using reversed-phase high-performance liquid chromatography technique.[1][2]
These Secondary Standards are qualified as Certified Reference Materials. These are suitable for use in several analytical applications including but not limited to pharma release testing, pharma method development for qualitative and quantitative analyses, food and beverage quality control testing, and other calibration requirements.
Biochem/physiol Actions
Broad-spectrum anticonvulsant.
General description
Effective therapy for partial seizures in adults and childhood seizures in Lennox-Gastaut syndrome.
Rufinamide is an antiepileptic drug, used as an adjunctive for the treatment of seizures associated with Lennox−Gastaut syndrome.[1] Its mode of action involves prolonging the refractory period of voltage-dependent sodium channels via stabilization of the sodium channel inactive state, thus effectively keeping the ion channels closed.[1]
Other Notes
This Certified Reference Material (CRM) is produced and certified in accordance with ISO 17034 and ISO/IEC 17025. All information regarding the use of this CRM can be found on the certificate of analysis.
To see an example of a Certificate of Analysis for this material enter LRAA5294 in the slot below. This is an example certificate only and may not be the lot that you receive.
Rufinamide
Pharmaceutical Secondary Standard; Certified Reference Material
Code: PHR1625
- Manufacturer: Merck
- Trademark: Supelco
- Hãng sản xuất: Merck
Kiểm tra giá Rufinamide
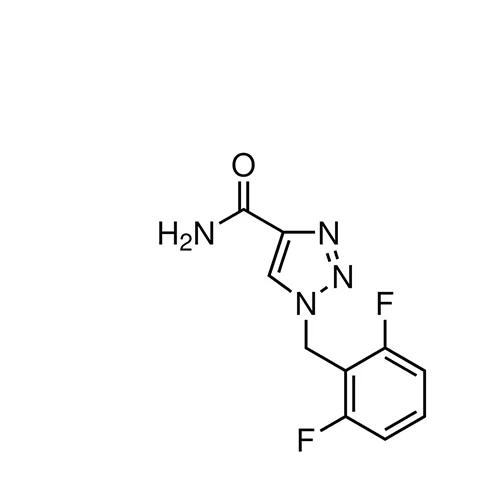
Rufinamide
Bạn vui lòng nhập đúng số điện thoại để chúng tôi sẽ gọi xác nhận đơn hàng trước khi giao hàng. Xin cảm ơn!


 Tiếng Việt
Tiếng Việt