Application
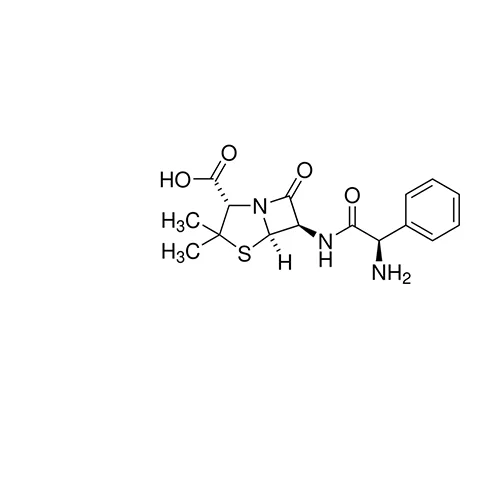
Anhydrous ampicillin BP Reference standard, intended for use in laboratory tests only as specifically prescribed in the British Pharmacopoeia.
Also used in monographs such as:
Co-fluampicil Oral Suspension
Co-fluampicil Capsules
Ampicillin Oral Suspension
Ampicillin Injection
Ampicillin Capsules
Ampicillin Trihydrate and Cloxacillin Benzathine Intramammary Infusion (Dry Cow)
Piperacillin Infusion
Ampicillin Sodium and Cloxacillin Sodium Intramammary Infusion (Lactating Cow)
Biochem/physiol Actions
Mode of Action: Ampicillin is a semisynthetic penicillin and a β-lactam antibiotic that inhibits bacterial cell-wall synthesis by inactivating transpeptidases on the inner surface of the bacterial cell membrane.
Mode of Resistance: Administration with β-lactamase cleaves the β-lactam ring of Ampicillin and inactivates it.
Antimicrobial Spectrum: Effective against both Gram-positive (similar to benzylpenicillin) and Gram-negative bacteria (similar to tetracyclines and chloramphenicol.
General description
This product is provided as delivered and specified by the issuing Pharmacopoeia. All information provided in support of this product, including SDS and any product information leaflets have been developed and issued under the Authority of the issuing Pharmacopoeia. For further information and support please go to the website of the issuing Pharmacopoeia.
Other Notes
Sales restrictions may apply.
Packaging
Unit quantity: 250 mg. Subject to change. The product is delivered as supplied by the issuing Pharmacopoeia. For the current unit quantity please visit British Pharmacopoeia


 Tiếng Việt
Tiếng Việt