Application
Dienogest for system suitability EP Reference standard, intended for use in laboratory tests only as specifically prescribed in the European Pharmacopoeia.
Biochem/physiol Actions
Dienogest is a fourth-generation progestin, an orally active synthetic progestogen with approximately 10% of the relative affinity of progesterone, but has a very potent progestagenic effect in the endometrium with some antiandrogenic activity and without any androgenic, glucocorticoid or mineralocorticoid effects. It has been used alone or in combination as a contraceptive and to treat uterine fibroids. Dienogest inhibits ovulation primarily through peripheral action rather than by central action on gonadotrophin secretion.
Dienogest is an orally active synthetic progestogen
General description
This product is provided as delivered and specified by the issuing Pharmacopoeia. All information provided in support of this product, including MSDS and any product information leaflets have been developed and issued under the Authority of the issuing Pharmacopoeia.
For further information and support please go to the website of the issuing Pharmacopoeia.
Other Notes
Sales restrictions may apply.
Packaging
The product is delivered as supplied by the issuing Pharmacopoeia. For the current unit quantity, please visit the EDQM reference substance catalogue.
Dienogest for system suitability
European Pharmacopoeia (EP) Reference Standard
Code: Y0001782
- Manufacturer: Merck
- Trademark: Supelco
- Hãng sản xuất: Merck
Kiểm tra giá Dienogest for system suitability
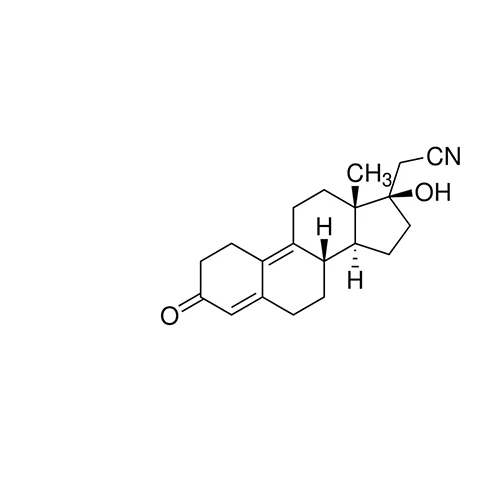
Dienogest for system suitability
Bạn vui lòng nhập đúng số điện thoại để chúng tôi sẽ gọi xác nhận đơn hàng trước khi giao hàng. Xin cảm ơn!


 Tiếng Việt
Tiếng Việt