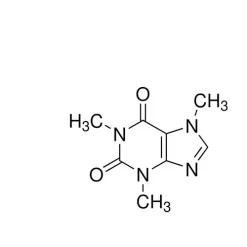
General description
Pharmaceutical secondary standards for application in quality control provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards
Analysis Note
These secondary standards offer multi-traceability to the USP and EP primary standards, where they are available.
Other Notes
This Certified Reference Material (CRM) is produced and certified in accordance with ISO 17034 and ISO/IEC 17025. All information regarding the use of this CRM can be found on the certificate of analysis.
To see an example of a Certificate of Analysis for this material enter LRAB3286 in the Documents slot below. This is an example certificate only and may not be the lot that you receive.
Gabapentin Impurity G
Pharmaceutical Secondary Standard; Certified Reference Material
Code: PHR1979
- Manufacturer: Merck
- Trademark: Supelco
- Hãng sản xuất: Merck
Kiểm tra giá Gabapentin Impurity G

Gabapentin Impurity G
Bạn vui lòng nhập đúng số điện thoại để chúng tôi sẽ gọi xác nhận đơn hàng trước khi giao hàng. Xin cảm ơn!


 Tiếng Việt
Tiếng Việt