Application
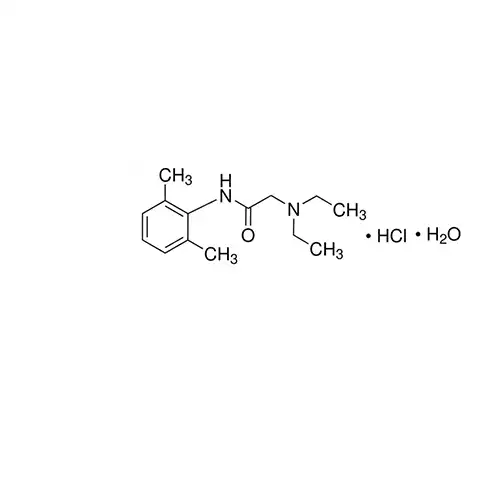
Lidocaine hydrochloride BP Reference standard, intended for use in laboratory tests only as specifically prescribed in the British Pharmacopoeia.
Also used in monographs such as:
Bupivacaine and Adrenaline Injection / Bupivacaine and Epinephrine Injection
Lidocaine and Adrenaline Injection / Lidocaine and Epinephrine Injection
Mepivacaine Injection
Lidocaine Intraocular Injection
Biochem/physiol Actions
Na+ channel blocker; class IB antiarrhythmic that is rapidly absorbed after parenteral administration
Na+ channel blocker; class IB antiarrhythmic.
General description
This product is provided as delivered and specified by the issuing Pharmacopoeia. All information provided in support of this product, including SDS and any product information leaflets have been developed and issued under the Authority of the issuing Pharmacopoeia. For further information and support please go to the website of the issuing Pharmacopoeia.
Other Notes
Sales restrictions may apply.
Learn how British Pharmacopoeia (BP) sets the standard by offering quality British Pharmacopoeia Chemical Reference Substances (BPCRS).
Packaging
Unit quantity: 100 mg. Subject to change. The product is delivered as supplied by the issuing Pharmacopoeia. For the current unit quantity please visit British Pharmacopoeia


 Tiếng Việt
Tiếng Việt