General description
This product is provided as delivered and specified by the issuing Pharmacopoeia. All information provided in support of this product, including SDS and any product information leaflets have been developed and issued under the Authority of the issuing Pharmacopoeia. For further information and support please go to the website of the issuing Pharmacopoeia.
Application
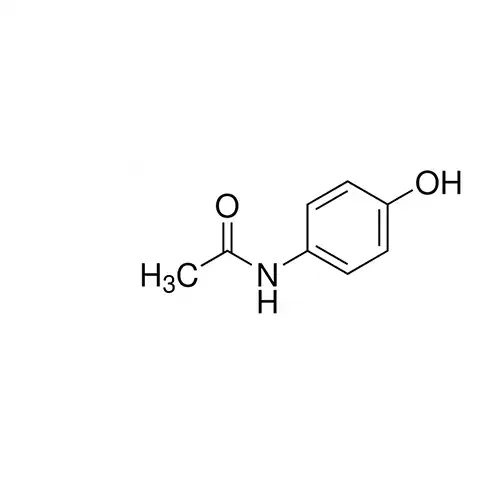
Analgesic.
Paracetamol BP Reference standard, intended for use in laboratory tests only as specifically prescribed in the British Pharmacopoeia.
Also used in monographs such as:
Paracetamol, Codeine Phosphate and Caffeine Capsules
Co-dydramol Tablets
Co-codamol Capsules
Co-codamol Tablets
Co-codamol Effervescent Tablets
Paracetamol, Codeine Phosphate and Caffeine Tablets
Paracetamol and Caffeine Soluble Tablets
Paracetamol and Caffeine Tablets
Paracetamol Suppositories
Paracetamol Soluble Tablets
Paracetamol Effervescent Tablets
Paracetamol Capsules
Paracetamol Oral Suspension
Paediatric Paracetamol Oral Solution
Paediatric Paracetamol Oral Suspension
Paracetamol Tablets
Paracetamol Dispersible Tablets
Packaging
Unit quantity: 100 mg. Subject to change. The product is delivered as supplied by the issuing Pharmacopoeia. For the current unit quantity please visit British Pharmacopoeia
Other Notes
Learn how British Pharmacopoeia (BP) sets the standard by offering quality British Pharmacopoeia Chemical Reference Substances (BPCRS).
Sales restrictions may apply.


 Tiếng Việt
Tiếng Việt