Application
Pioglitazone for system suitability EP Reference standard, intended for use in laboratory tests only as specifically prescribed in the European Pharmacopoeia.
Biochem/physiol Actions
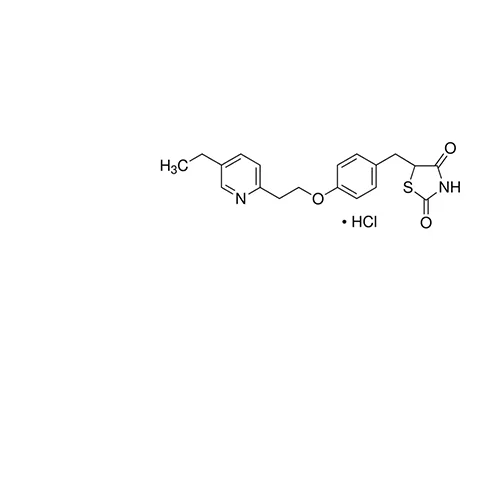
Pioglitazone hydrochloride is a PPARγ agonist and thiazolidinedione (TZD) anti-diabetic.
Pioglitazone hydrochloride is a PPARγ agonist and thiazolidinedione (TZD) anti-diabetic. Pioglitazone is a selective agonist of the nuclear receptor peroxisome proliferator-activated receptor γ (PPAR-γ) and to a lesser extent PPAR-α.
General description
This product is provided as delivered and specified by the issuing Pharmacopoeia. All information provided in support of this product, including SDS and any product information leaflets have been developed and issued under the Authority of the issuing Pharmacopoeia.For further information and support please go to the website of the issuing Pharmacopoeia.
Other Notes
Sales restrictions may apply.
Packaging
The product is delivered as supplied by the issuing Pharmacopoeia. For the current unit quantity, please visit the EDQM reference substance catalogue.
Pioglitazone for system suitability
European Pharmacopoeia (EP) Reference Standard
Code: Y0001924
- Manufacturer: Merck
- Trademark: Supelco
- Hãng sản xuất: Merck
Kiểm tra giá Pioglitazone for system suitability
Pioglitazone for system suitability
Bạn vui lòng nhập đúng số điện thoại để chúng tôi sẽ gọi xác nhận đơn hàng trước khi giao hàng. Xin cảm ơn!


 Tiếng Việt
Tiếng Việt