General description
This Certified Reference Material (CRM) is produced and certified in accordance with ISO 17034 and ISO/IEC 17025. All information regarding the use of this CRM can be found on the certificate of analysis.
Pharmaceutical secondary standards for application in quality control provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards.
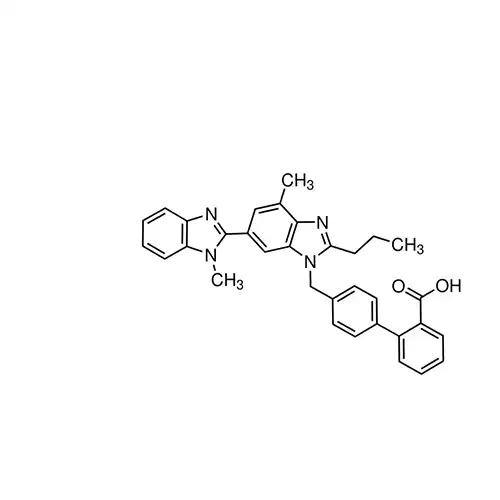
Telmisartan belongs to the class of angiotensin II receptor antagonists (ARAs) used in the treatment of hypertension.
Application
This pharmaceutical secondary standard can also be used as follows:
Reversed-phase high-performance liquid chromatography (RP-HPLC) method-based multi-analysis of rosuvastatin, telmisartan, ezetimibe, and atorvastatin in pharmaceutical dosage formulations[1]
RP-HPLC analysis of telmisartan in the presence of its three impurities in its tablet formulation following an analytical quality-by-design (AQbD) approach[2]
Multi-determination of hydrochlorothiazide, amlodipine, and telmisartan by HPLC combined with spectrometric techniques without derivatization
Simultaneous estimation of telmisartan and hydrochlorothiazide by micellar high-pressure liquid chromatography and spectrophotometry-based methods in their combined tablet formulations[3]
RP-HPLC method-based determination of four cardiovascular drugs—atorvastatin, simvastatin, telmisartan, and irbesartan in bulk drugs and tablet dosage forms, following ICH guidelines
Biochem/physiol Actions
Telmisartan is a non-peptide AT1 angiotensin receptor antagonist.
Analysis Note
These secondary standards offer multi-traceability to the USP and EP primary standards, where they are available.
Other Notes
To see an example of a Certificate of Analysis for this material enter LRAC3179 in the Documents slot below. This is an example certificate only and may not be the lot that you receive.


 Tiếng Việt
Tiếng Việt