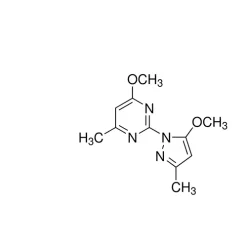
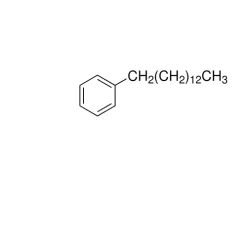
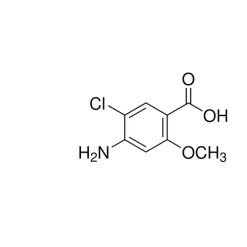
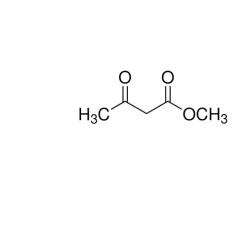
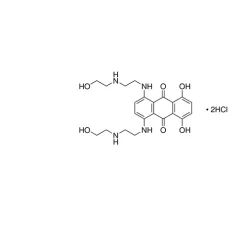
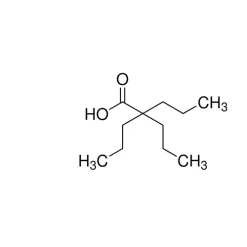
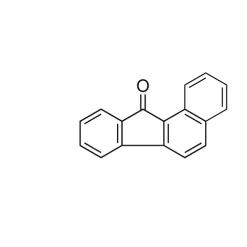
General description
A unit of NIST SRM 909C human serum consists of three vials, each containing approximately 2 mL of frozen human serum.
SRM 909C_Cert
SRM 909C_SDS
Application
The human blood serum standard reference material (SRM) is meant primarily for use in validating analytical methods for the determination of specified constituents in human serum. This SRM can also be used for quality assurance when assigning values to in-house control materials.
Features and Benefits
The certified values are traceable to the International System of Units (SI) for the amount of substance and mass, expressed both in mass concentrations (milligrams per deciliter, micrograms per deciliter, nanograms per milliliter, or grams per liter) and amount concentration (nanomoles per liter or micromoles per liter)
Analysis Note
Endotoxin tested
Other Notes
Certified for the analytes listed below.
Cholesterol, Creatinine, Glycerides, Urea, Calcium (Ca), Chloride (Cl), Magnesium (Mg), Potassium (K), Sodium (Na), Iron (Fe), Selenium (Se), Transferrin (Tf)
Reference/Informational value is provided for the following analytes.
Total Protein, Disialo Tf, Trisialo Tf, Tetrasialo Tf, Pentasialo Tf, Hexasialo Tf
See certificate for values and more details at nist.gov/SRM.
Refer to the certificate for information on expiry, safety, usage, and storage.
Notes on biomaterials, disposal, and transport information are available in SDS.
Legal Information
NIST is a registered trademark of National Institute of Standards and Technology
SRM is a registered trademark of National Institute of Standards and Technology
Disclaimer
RESEARCH USE ONLY. This product is regulated in France when intended to be used for scientific purposes, including for import and export activities (Article L 1211-1 paragraph 2 of the Public Health Code). The purchaser (i.e. enduser) is required to obtain an import authorization from the France Ministry of Research referred in the Article L1245-5-1 II. of Public Health Code. By ordering this product, you are confirming that you have obtained the proper import authorization.


 English
English