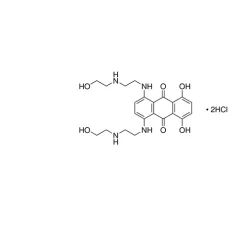
General description
may contain traces of (NH4)2SO4 and sodium phosphateL6010
Application
α-Lactalbumin from bovine milk has been used:
in indirect enzyme-linked immunosorbent assay (ELISA) and competitive ELISA methods[1]
in binding of Hsp90 and HSJ1b analysis[2]
to inhibit the lysozyme CAP-RAST assay[3]
Biochem/physiol Actions
α-Lactalbumin is the cheif protein in human milk. It consists of a single polypeptide chain with 8 cysteines which form disulfide bridges. α-Lactalbumin binds several metal ions, including calcium, which is thought to play a role in the regeneration of native α-lactalbumin from the reduced, denatured form.[4] α-Lactalbumin also has a distinct zinc binding site that is thought to play a role in the binding of the lactose synthase complex.[5][6]The mature protein consists of 123 amino acid residues (14 kD), and it has a three-dimensional structure with 1.7 Angstrom resolution, demonstrating four α-helices and a triple stranded antiparallel β-sheet.[7]
Alters the substrate specificity of galactosyltransferase to increase the rate of lactose formation; the complex of galactosyltransferase and α-lactalbumin is called lactose synthase.
Alters the substrate specificity of galactosyltransferase to increase the rate of lactose formation; the complex of galactosyltransferase and α-lactalbumin is called lactose synthase. Site-directed mutagenesis of Asp87 or Asp88 to Ala completely abolishes the strong calcium binding affinity and reduces the stimulation of lactose synthase to <3.5% of the maximal rate.
Analysis Note
May contain traces of (NH4)2SO4 and sodium phosphate.


 English
English