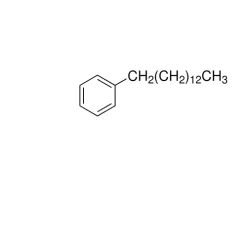
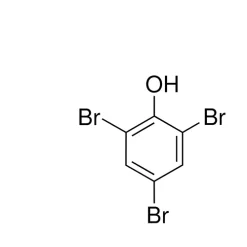
General description
Retinol Binding Protein (RBP) is a member of lipocalin superfamily.[1] The normal concentration of RBP in serum is 40 to 50 μg/mL and has a molecular weight of 21 kDa. It has a turnover rate of 4 to 24 h.[2] RBP is produced in the liver in response to retinol binding trigger. It is also produced in kidney, sertoli cells of testis.[1] RBP contains a single globular domain and a highly conserved N-terminal region. It has eight antiparallel β sheets in the centre which binds retinol.[3] In human chromosome, the gene encoding RBP is localised at 3q23.[4]
Application
Retinol Binding Protein from human urine has been used:
as one of the protein in microinjecting mesencephalic cavity of chicken eggs to monitor embryonic cerebrospinal fluid/embryonic serum protein transfer[5]
as an additive in basal medium for RNA transfection experiments[6]
in the antibody-derivatized affinity pipettes preparation for the capture of affinity proteins[7]
Biochem/physiol Actions
Inflammation induced by lipopolysaccharide (LPS) exposure caused a reduction in the level of retinol binding protein produced by the liver. [8]
Retinol Binding Protein (RBP) is a specific protein for the transport of vitamin A. The apo-RBP, formed after delivering retinol to the tissues is catabolized by the kidney. The plasma concentration of RBP is less in diabetes mellitus patients.[2] RBP binds to the carrier protein transerythrin, to prevent its loss during glomerular filtration.[1] RBP is implicated in initial neurogenesis and neuroepithelial cell proliferation.[5] RBP is responsible for blepharophimosis syndrome (BPES).[4]
Physical form
Solution in 10 mM sodium phosphate, pH 7.4, 150 mM NaCl, and 0.05% NaN3


 English
English