General description
This product is provided as delivered and specified by the issuing Pharmacopoeia. All information provided in support of this product, including SDS and any product information leaflets have been developed and issued under the Authority of the issuing Pharmacopoeia.For further information and support please go to the website of the issuing Pharmacopoeia.
Application
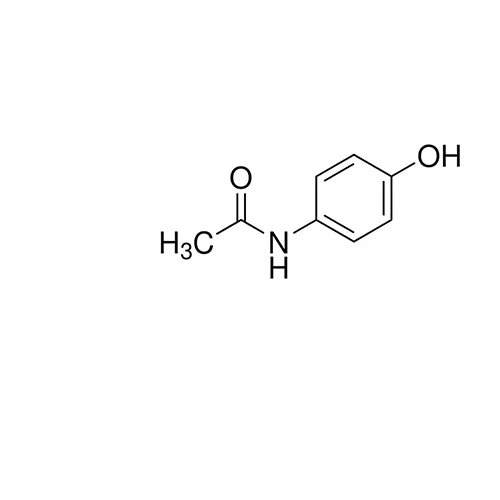
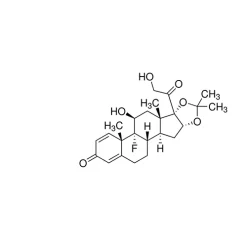
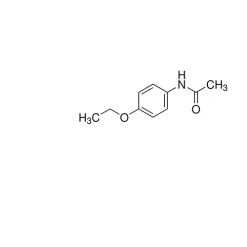
Acetaminophen USP reference standard, intended for use in specified quality tests and assays as specified in the USP compendia.
Also, for use with USP monographs such as:
Acetaminophen Tablets
Acetaminophen Capsules
Acetaminophen Suppositories
Acetaminophen Oral Suspension
Acetaminophen Oral Solution
Acetaminophen and Caffeine Tablets
Acetaminophen Extended-Release Tablets
Analgesic.
Analysis Note
These products are for test and assay use only. They are not meant for administration to humans or animals and cannot be used to diagnose, treat, or cure diseases of any kind.
Other Notes
Sales restrictions may apply.


 English
English